
Learn about the elements of the periodic table and how they are organized.
This article was last updated August 28th, 2024.
Matter that is composed of only one type of atom is called an element. People have known about some elements such as gold, silver, iron, mercury and tin for hundreds of years. Scientists discovered many others during the 18th and 19th centuries. Some elements have even been discovered within your lifetime!
Many of the elements have their name or their symbol based in Latin or Greek. For example:
| Element | Atomic Symbol | Word origin |
|---|---|---|
| Gold | Au | Latin word aurum meaning ‘shining dawn’ |
| Mercury | Hg | Greek word hydrargyrum meaning ‘silver water’ |
| Chlorine | Cl | Greek word chloros meaning ‘green’ |
| Neon | Ne | Greek word neos meaning ‘new’ or ‘different’ |
As well for famous scientists such as:
| Element | Atomic Symbol | Word origin |
|---|---|---|
| Curium | Cm | Pierre and Marie Curie (research in radioactivity) |
| Einsteinium | Es | Albert Einstein |
| Nobelium | No | Alfred Nobel (of Nobel Prize fame) |
| Mendelevium | Md | Dmitri Mendeleev (creator of the first periodic table) |
Other elements have been named for places, such as:
| Element | Atomic Symbol | Word origin |
|---|---|---|
| Europium | Eu | Europe |
| Francium | Fr | France |
| Germanium | Ge | Germany |
| Berkelium | Bk | Berkeley, California, USA |
| Dubnium | Db | Dubna, Russia |
Did you know?
The country Argentina got its name from the element silver. Argentum is the Latin name for silver.
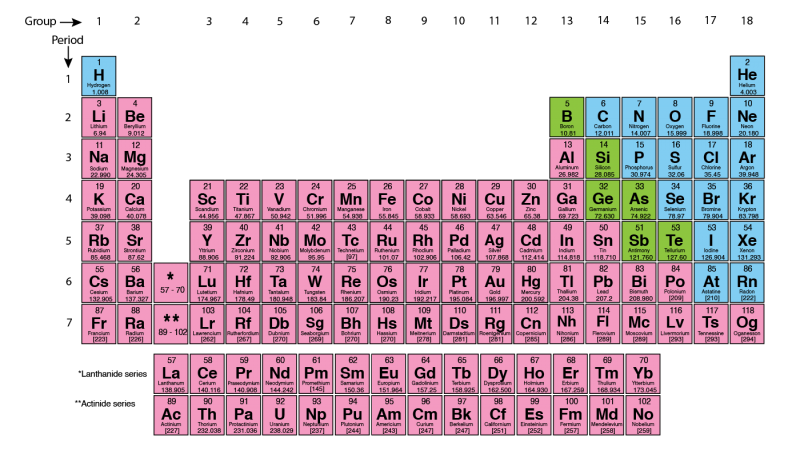
The periodic table of the elements is a visual and logical way to organize all elements. Dmitri Mendeleev, a Russian scientist, is usually credited with creating the first periodic table in 1869. In Germany, Lothar Meyer also came up with an almost identical table later the same year. Some people attribute the first categorization to Alexandre-Emile Béguyer de Chancourtois.
Click here to access screen reader Accessible Periodic Tables
Each element on the periodic table is listed in a box with its atomic symbol and atomic number. The element’s full name and atomic mass is also sometimes indicated. The image below shows a typical entry for the element calcium.
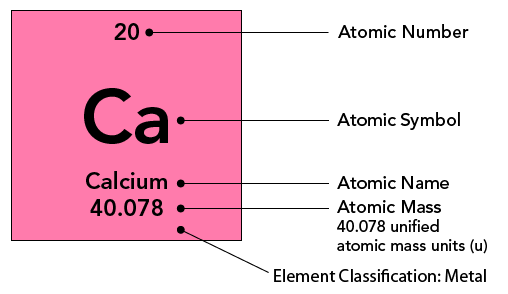
Image - Text Version
Shown is a labelled diagram of the calcium square from the periodic table.
The square is pink with four centred lines of black text. The pink area is labelled “Element Classification: Metal.”
Starting at the top, the first line reads, “20” in small, unbolded font. This is labelled “Atomic Number.”
Below, the second line is in the centre of the square, in a much larger and bold font. it reads, “Ca.” This is labelled “Atomic Symbol.”
The third line is in smaller and unbolded text. It reads, “Calcium.” This is labelled “Atomic Name.”
The last line is also small and unbolded. It reads, “40.078.” This is labelled “Atomic Mass, 40.078 unified atomic mass units (u).”
The number above the atomic symbol represents the atomic number. The atomic number is equal to the number of protons in an atom's nucleus and determines which element an atom is. For example, any atom that contains exactly 20 protons in its nucleus is an atom of calcium. The number of protons influences the chemical behaviour of an element.
The atomic mass of an element refers to the average mass of an element in unified atomic mass units (short form u). This is usually given at the bottom of an element’s entry in the periodic table. The atomic mass is a decimal number because it is an average of the masses of the various isotopes of an element. Isotopes of an element have the same number of protons, but different numbers of neutrons. For example, hydrogen has three naturally occurring isotopes. The most common isotope, protium, has no neutrons! Deuterium has one neutron and tritium has two neutrons.

Image - Text Version
Shown is a colour diagram of protium, deuterium and tritium.
The title reads, “Main Hydrogen Atom Isotopes.” Below are three separate illustrations.
Starting on the left, the first illustration is labelled, “Protium (1H).” It shows a small blue circle with a negative symbol moving in an oval orbit around a larger red circle with a positive sign. The blue circle is labelled “Electron, Negatively charged particle.” The red circle is labelled “Proton, Positively charged particle.”
In the middle, the second illustration is labelled “Deuterium (2H).” This is the same as the first, except one orange circle is attached to the proton. This is labelled “Neutron, Particle with no charge.”
On the right, the third illustration is labelled “Tritium (3H).” This is the same as the others, except two neutrons are attached to the proton.
Scientists have also created other highly unstable isotopes of hydrogen. Those, with up to 7 neutrons, do not occur in nature.
Getting the average number of neutrons for an element is easy. You need to subtract the number of protons (atomic number) from the atomic mass. For example, calcium has an atomic number of 20 and an atomic mass of 40.078 u. By subtracting 20 from 40 (rounded), we determine that the average number of neutrons is 20.
Mendeleev organized the table so that the elements from low to high atomic mass. Then he organized rows and columns to highlight elements with similar chemical properties. The rows are known as periods. All elements in the same period have the same number of electron layers, also called shells. The columns are known as groups (or families).
Mendeleev also left gaps in his table. He believed that some elements had not yet been discovered. Those gaps were for undiscovered elements. Mendeleev predicted the missing elements would have the same properties as the other elements in the same column.
Discoveries led to many new elements during the 1900s. Mendeleïev’s table was rearranged to give us the format that we use today. But the principles of groups and periods still exist in our modern version.
The groups on the periodic table are numbered from 1 to 18. The elements within a group usually have similar properties. For example, the elements of group 18 are all gases that do not readily react with other elements. Groups can sometimes be given names. For example, the elements in group 18 are called ‘Noble Gases’.
Did you know?
Noble gases get their name because the ability to stay calm and not react is considered noble in humans!

Image - Text Version
Shown is a vertical column of six blue squares from the periodic table.
The top square contains the atomic number 2, the atomic symbol He, the atomic name Helium, and the atomic mass 4.003.
The second square contains the atomic number 10, the atomic symbol Ne, the atomic name Neon, and the atomic mass 20.180.
The third square contains the atomic number 18, the atomic symbol Ar, the atomic name Argon, and the atomic mass 39.948.
The fourth square contains the atomic number 36, the atomic symbol Kr, the atomic name Krypton, and the atomic mass 83.798.
The fifth square contains the atomic number 54, the atomic symbol Xe, the atomic name Xenon, and the atomic mass 131.293.
The sixth square contains the atomic number 86, the atomic symbol Rn, the atomic name Radon, and the atomic mass [222].
Except for hydrogen, elements on the left side of the periodic table are metals. Metals are solid at room temperature, except for mercury, which is liquid. They are also malleable and can be ductile. Ductile is another word for stretchy. Metals also have a shiny appearance. They are good conductors of heat and electricity.
Elements on the right side of the periodic table are non-metals. Non-metals can be solids, liquids or gases at room temperature. They are poor conductors of heat and electricity.
Elements that have some metallic as well as non-metallic properties are known as metalloids (e.g., silicon, arsenic, etc.).
We sometimes call the elements in groups 3 to 12 the d-block. These elements are also known as transition elements.

Image - Text Version
Shown are four rows of pink squares from the periodic table, arranged in a grid. The top two rows have ten squares, the bottom two rows have nine.
The first row includes elements with atomic numbers from 21-30. The second row includes atomic numbers from 39-48. The third row starts with one square of blank space. Then it goes from numbers 72-80. The fourth row also starts with one square of blank space. Then it goes from 104-112. Please see accessible periodic tables for full details.
There are two rows placed at the bottom of the periodic table. These are the Lanthanide and Actinide series. The Lanthanide series includes fifteen metallic elements with atomic numbers 57 through 71. The Lanthanide elements, along with the two elements of group 3, Scandium and Yttrium, are often called the rare earth elements. You can find Lanthanides naturally on Earth. The Actinide series is much different. All of the Actinide elements are radioactive and some are not found in nature. Some of the elements with higher atomic numbers can only be created in laboratories.

Image - Text Version
Shown are two rows of pink squares from the periodic table, arranged in a grid.
The top row is labelled “Lanthanides” and goes from atomic number 57-71. The bottom row is labelled “Actinides” and goes from atomic number 89-103. Please see accessible periodic tables for full details.
In general, the size or radius of an atom increases as you move down a group. As you go down, elements have more layers of electrons, which results in bigger atoms. From left to right within a period, atomic size generally decreases. This is because more protons pull the electrons closer to the nucleus.
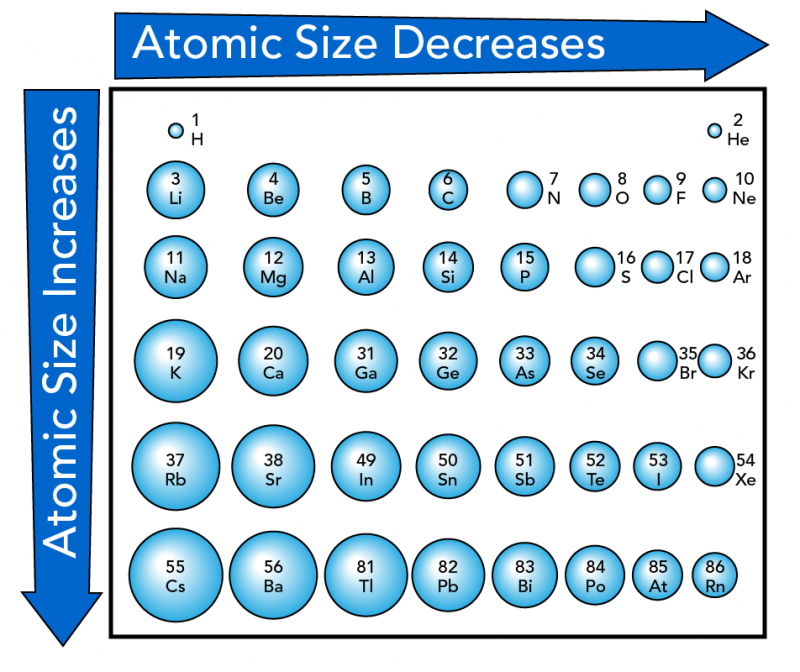
Image - Text Version
Shown is a colour chart of atoms shown as different sized blue spheres, labelled with atomic numbers and symbols.
The spheres are arranged in a grid of six rows and eight columns. A large blue arrow across the top points from left to right. It reads, “Atomic Size Decreases.” Another blue arrow along the left edge points from top to bottom. It reads “Atomic Size Increases.” The spheres below follow these patterns. Each one in the row is smaller than the last from left to right. And each one is larger than the last in the column from top to bottom. The smallest sphere is labelled “2, He” on the top right. The largest is labelled “55, Cs” on the lower left.
The top row contains only two spheres. A small one on the far left is labelled “1, H.” An even smaller one on the far right is labelled “2, He.” The next row starts with “3, Li” on the left and goes to “10, Ne” on the right. The third starts with “11, Na” on the left and goes to “18, Ar” on the right. The fourth starts with “19, K” and goes to “36, Kr”. The fifth starts with “37, Rb” and goes to 54 Xe”. The sixth starts with “55, Cs” and goes to “86, Rn”.
Starting on the left, the first column goes from “1, H” at the top to “55, Cs” at the bottom. The second goes from “4, Be” at the top to “56, Ba” at the bottom. The third goes from “5, B” to “81, Ti”. The fourth goes from “6, C” to “82, Pb”. The fifth goes from “7, N” to “83, Bi”. The sixth goes from “8, O” to “84, Po”. The seventh goes from “9, F” to “85, At”. The eighth goes from “2, He” to *6, Rn”.
Electronegativity is the chemical property that describes the tendency of an atom to form bonds with others. Electronegativity usually decreases down each group and increases from left to right across a period. The most electronegative elements are in the upper right hand side of the periodic table. This is because non-metals have high electronegativity. They tend to attract electrons from other atoms. While metals tend to lose electrons easily.
To become electrically-charged, stable atoms must lose or gain electrons. There are two types of electric charges. There are positive charges (+) and negative charges (-). Substances with the same charge will repel each other. While substances with opposite charges attract each other. This is similar to how magnets work!
The atoms of some elements can lose or gain electrons more easily than others. For example, the elements in group 1 (e.g., Li, Na, K) can easily lose one electron, which gives them a +1 charge. The elements in group 17 (Cl, Br, I) can easily gain one electron, which gives them a -1 charge. When an atom or molecule has a charge, it is said to be an ion. When an atom or molecule has more electrons than protons, the result is a negative ion, called an anion. When it has fewer electrons than protons, then the result is a positive ion, called a cation.
Each group in the periodic table has a specific charge commonly associated with its ions. The exceptions to this are the transition elements, and the lanthanide and actinide series. Below are the charges for the different groups within the table.
| Group | 1 | 2 | 3 | Transition elements, Lanthanide and Actinide series | 15 | 16 | 17 | 18 |
|---|---|---|---|---|---|---|---|---|
| Charge | +1 | +2 | +3 | Various | -3 | -2 | -1 | 0 |
You may have noticed that the elements in group 18 have a charge of 0. This is because these elements are very stable and they do not readily gain or lose electrons.
This backgrounder was lasted updated on: August 28th, 2024
The photographic periodic table
This page by periodictable.com has photographs of common things made from, or using, each of the elements.
Introduction to the Particle Theory of Matter
This article from Let’s Talk Science introduces the history of the theory that describes matter as different assemblies of smaller particles, or atoms.
The Genius of Mendeleev's Table
This article from Let’s Talk Science presents how Dmitri Mendeleev created the first periodic table.
Introduction to the Atom
This article from Let’s Talk Science presents the history of the different atomic models created.
The Newest Elements on the Periodic Table
This article from Let’s Talk Science presents the most recent elements added to the periodic table.
The Periodic Table of Elements
This page by the Thomas Jefferson National Accelerator Facility has an interactive periodic table as well as flash cards, games, etc. about the elements.